|
Pritchard J, Han R, Horst N, Cruikshank WW, Smith TJ.Autoantibodies to IGF-1 binding sites in thyroid associated ophthalmopathy. Weightman DR, Perros P, Sherif IH, Kendall-Taylor P.Paper presented at: 48 th Annual Meeting of the North American Neuro-Ophthalmology Society (NANOS) February 12-17, 2022 Austin, TX. Sustainability of Teprotumumab Efficacy in Thyroid Eye Disease. Teprotumumab for the treatment of active thyroid eye disease. Douglas RS, Kahaly GJ, Patel A, et al.Teprotumumab for thyroid-associated ophthalmology. Poster session presented at: 48th Annual Meeting of the North American Neuro-Ophthalmology Society (NANOS) February 12-17, 2022 Austin, TX. Post-marketing Surveillance of Hearing-related Adverse Events in Patients with TED Treated With Teprotumumab. Graves’ Ophthalmopathy: VISA versus EUGOGO classification, assessment, and management. Barrio-Barrio J, Sabater AL, Bonet-Farriol E, Velázquez-Villoria A, Galofré JC.Horizon is focused on the discovery, development, and commercialization of medicines that address critical needs for people impacted by rare, autoimmune, and severe inflammatory diseases. We are committed to continuing our research to support a positive patient experience with this important medicine.” “Ongoing research reinforces the significant impact TEPEZZA can have on people living with this devastating disease. Sherman, MD, FACP, executive vice president, chief medical officer, Horizon. “Managing the disabling symptoms of Thyroid Eye Disease can be difficult for patients, both physically and emotionally, and before TEPEZZA, there was a significant unmet medical need for an effective treatment,” said Jeffrey W. 
Of those with data available, the majority of patients treated with TEPEZZA were proptosis, diplopia, and European Group on Graves’ Orbitopathy (EUGOGO) composite outcome responders up to 51 weeks after their last infusion. A total of 121 patients were analyzed, with 9 patients receiving additional medications and/or surgery during the follow-up periods. This speaks to the favorable benefit-risk profile of TEPEZZA for people living with Thyroid Eye Disease.”Ī separate analysis presented at NANOS 2022 examined the sustainability of response to TEPEZZA over the follow-up periods from the Phase 2 and OPTIC Phase 3 clinical trials and the OPTIC-X open-label extension study. “Notably, we continue to see high patient adherence to TEPEZZA, with more than 90% of patients completing their full course of treatment in the clinical studies and post-approval. “The majority of hearing-related adverse events in the pivotal trials and post-approval have been mild-to-moderate and reversible, but as with any medicine, it is important to monitor patients,” said Kimberly Cockerham, MD, adjunct clinical associate professor, Department of Ophthalmology, Stanford University School of Medicine, and a primary author of the report. Adverse events experienced in the clinical trials were manageable, with few discontinuations or therapy interruptions. 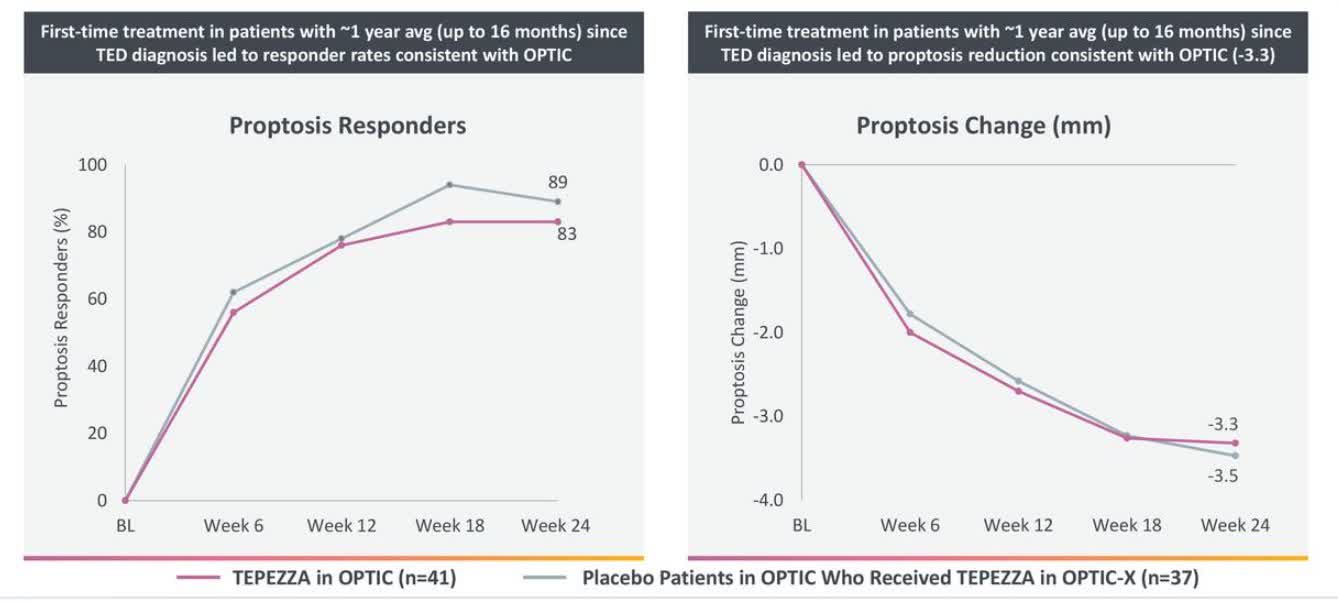
Hearing-related adverse events were reported in 9.5% (8/84) of patients treated with TEPEZZA, which were mild or moderate in severity, versus 0% of patients who received placebo. This is comparable with what was seen in the pooled OPTIC Phase 3 and OPTIC-X open-label extension trials for TEPEZZA, according to the company’s announcement. No new safety concerns were observed in the post-marketing database. 
The mean age of the patient, where known, was 59.3 years. The most frequently reported hearing event was hypoacusis (reduction in hearing), followed by tinnitus (ringing in the ears). 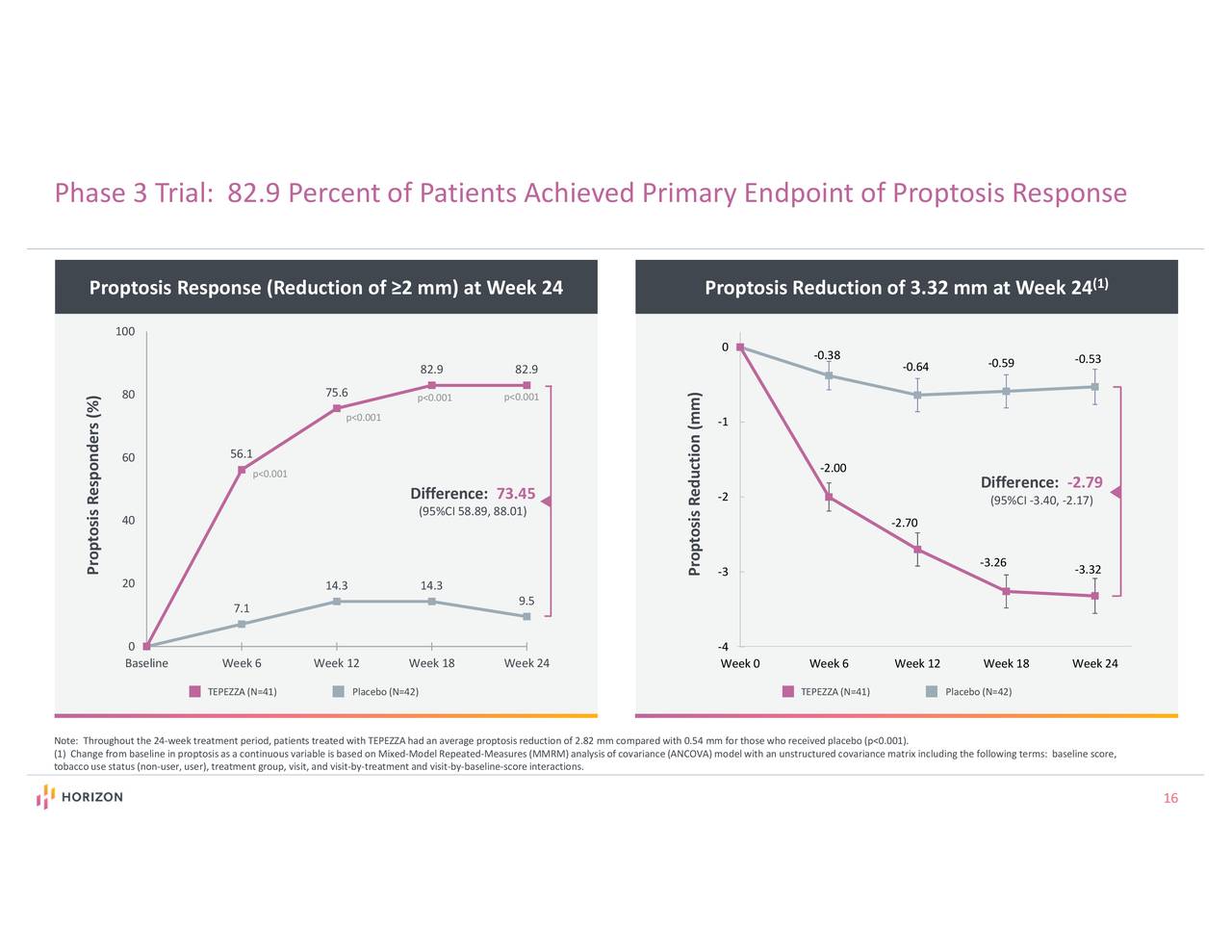
Among the thousands of patients included in this 19-month analysis (January 2020 – August 2021), approximately 10% of all cases reported to the safety database have included a hearing-related event. Since the FDA approval in 2020, Horizon has conducted post-marketing monitoring to understand the overall safety profile of TEPEZZA, including hearing-related events. Related article: Thyroid Eye Disease Drug May Cause Hearing Loss TEPEZZA is the “first and only medicine” approved by the US Food and Drug Administration (FDA) for the treatment of TED – a serious, progressive and potentially vision-threatening rare autoimmune disease. These findings, along with data on the sustainability of response to TEPEZZA, were presented at the 48 th Annual Meeting of the North American Neuro-Ophthalmology Society ( NANOS 2022), February 12-17, in Austin, Texas. Horizon Therapeutics announced results from a new post-marketing safety analysis of hearing events associated with TEPEZZA for the treatment of Thyroid Eye Disease (TED).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
